|
The valency of the compound is 1 and 1.Some formulas through which we can make compounds by knowing the valency of their elements are as follows: Hydrogen Chloride The electron configurations are written in the noble gas notation. This downloadable color periodic table contains each elements atomic number, atomic mass, symbol, name, and electron configuration. Hence, the valency of vanadium are 2,3,4 and 5.įormulas of Compounds by Valence Electrons Color Periodic Table With Electron Configurations.Its electronic configuration is Ar 3d 3 4s 2.It is a transition element and most of these show variable valences.It will gain 3 more electrons to complete its octet.Its electronic configuration is 2, 8, 5.It shows two valences: 1 (or Cu(I)), known as cuprous, and 2 (or Cu(II)), known as Cupric.It is a transition element, and most of these elements have variable valences.The outermost shell is already filled, so it will not lose electrons.Sodium will lose 1 electron to complete its octet.Its electronic configuration is 2, 8, 1.Here are the formulas of Valence Electrons for some elements: Sodium Read More: Polytetrafluoroethene (Teflon) The periodic table of all the elements is mentioned below: If there are 8 electrons in the outermost shell, it means that the octet is completed already, and there is no need to lose or gain electrons.If there are 4 electrons in the outermost shell, it can lose or gain 4 electrons to complete its octet.If there are 5, 6, or 7 electrons in the outermost shell, it will gain some electrons to complete its octet.If there are 1, 2, or 3 electrons in the outermost shell, it will lose these electrons to complete its octet.Another way to calculate the valence electron is by determining the electronic configuration of an element.įor Electronic configuration, we have to check the outermost shell of an electron in the following ways:.With the help of group numbers, we can easily calculate the number of valence electrons of an element.For a fully interactive version with orbitals, isotopes, compounds, and free printouts, visit. The periodic table shows the number of electrons in the valence shell of a chemical element that are available in the formation of a chemical bond. Valence electrons can be determined by checking the vertical column of the periodic table. Design Copyright © 2017 Michael Dayah (). 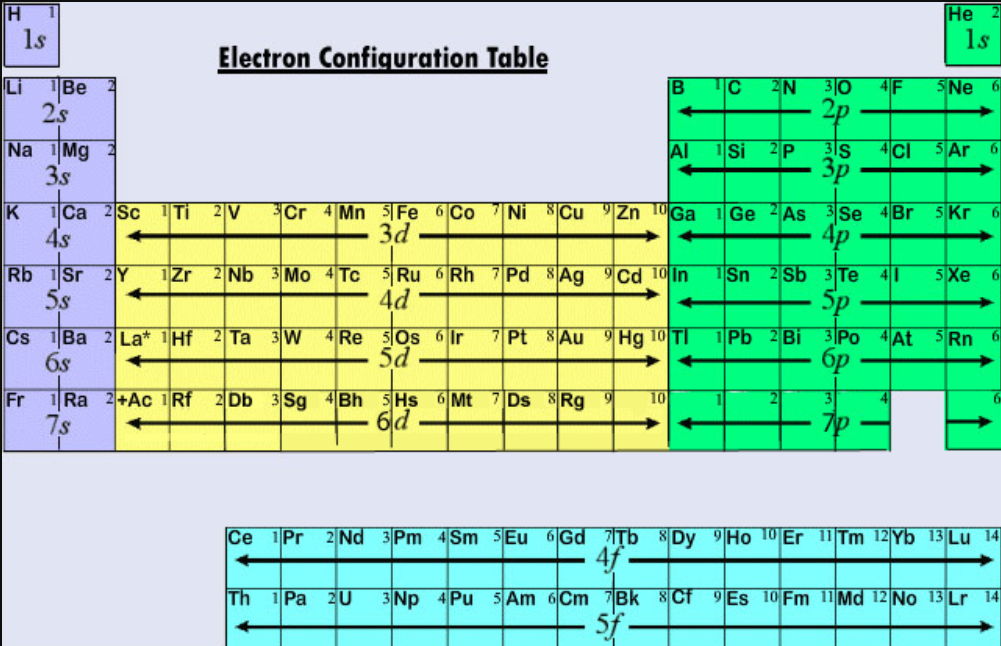
The table is arranged such that elements that have the same number of valence electrons fall under the same group. The structure of these elements could be much better. Explanation: The periodic table indicates the number of valence electrons an element has through its arrangement into groups and periods. The electrons of these groups can be determined easily, but they are difficult to predict. It can be found in all elements, even if they are from Group 3 to Group 12. This means that A has two valence electrons in 2s (2s 2) and five valence electrons in 2p (2p 5).Answer: 2s 2 2p 5. 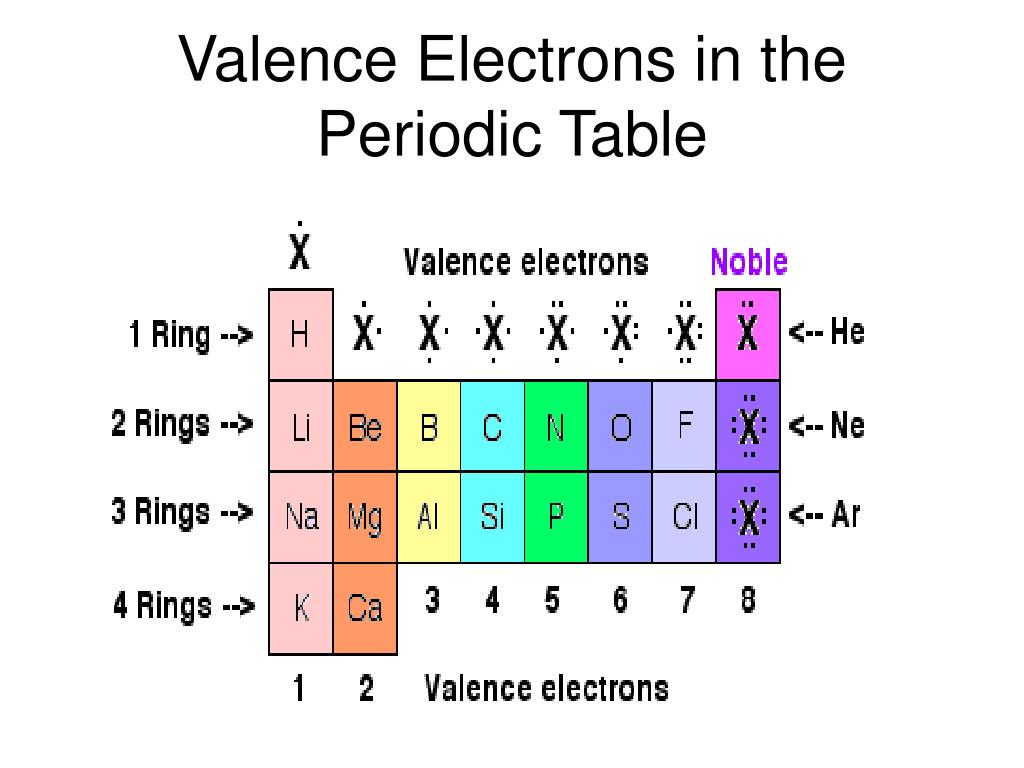
Valence electrons can easily be determined with the help of the periodic table. Element A is located in Period 2, the 5th position in 2p-block.Before the electrons are placed in 2p subshell, the 2s subshell must be filled first.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
